Bone defects are common and occur in many clinical situations including high grade open fractures with bone loss, high energy trauma, blast injuries, infection requiring debridement of bone, and resection of bone tumors or cysts.
There is a significant burden of disease associated with the management of bone defects, particularly if the bone defect is critical sized or when the planned reconstruction requires secondary surgery and autogenous bone grafting, often complicated by drawbacks such as donor site morbidity, limited graft volume, anesthesia time, need for additional surgical resources, and poor results in a significant number of patients.

❯ 01 Open and assemble
Open blister with the pre-filled syringe and the Tyvek pouch with the spindle drive (spindle nut and threaded spindle) and the PE-bag with the cannula. Push the spindle nut onto the syringe until a clear click sound is heard. Check that both sides of the spindle nut are locked.
❯ 02 Insert and adjust the spindle
Insert the threaded spindle into the spindle nut. Screw the threaded spindle in the spindle nut until it is in direct contact with the piston.

❯ 03 Adjust setting and application
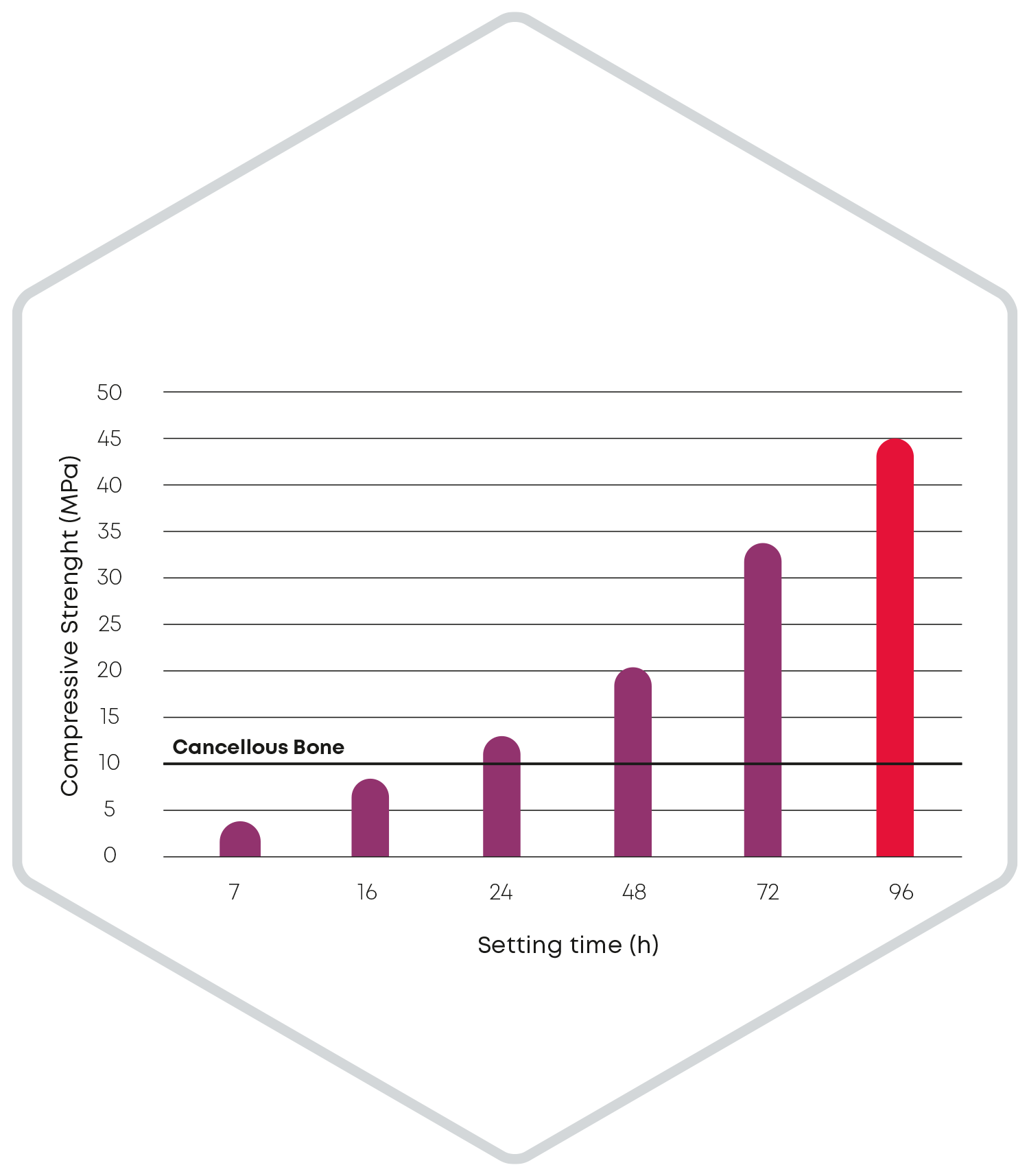
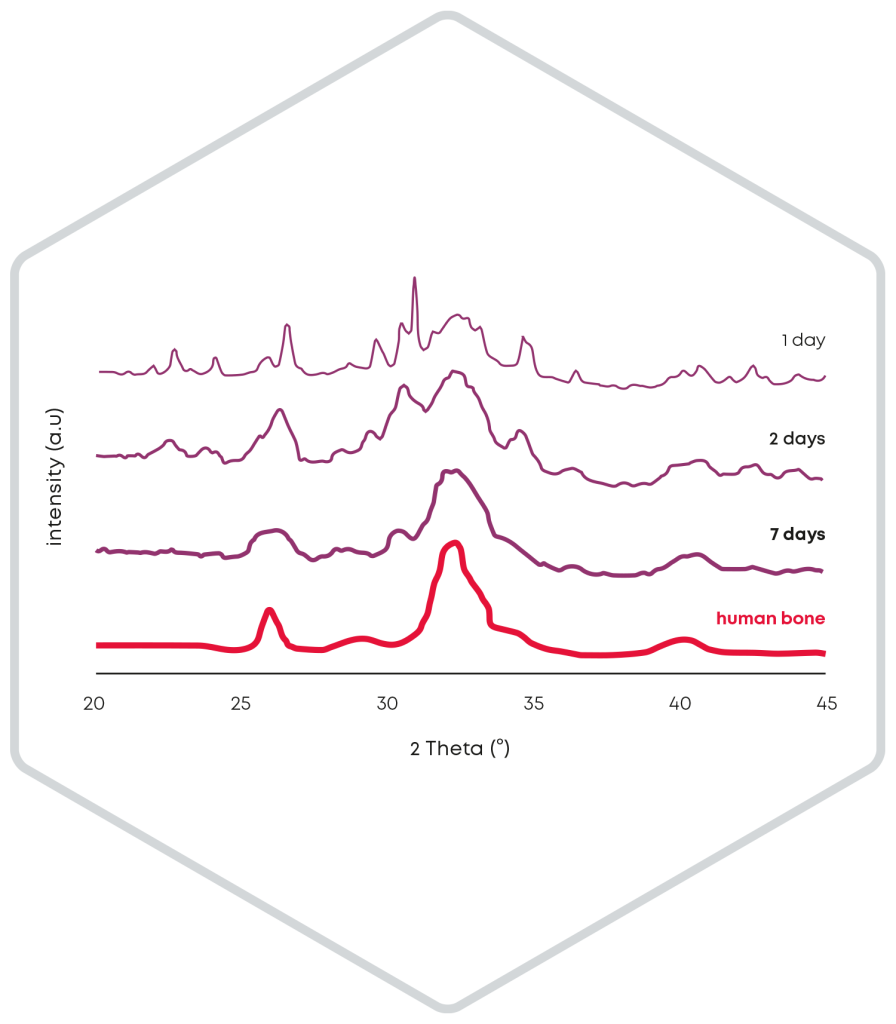
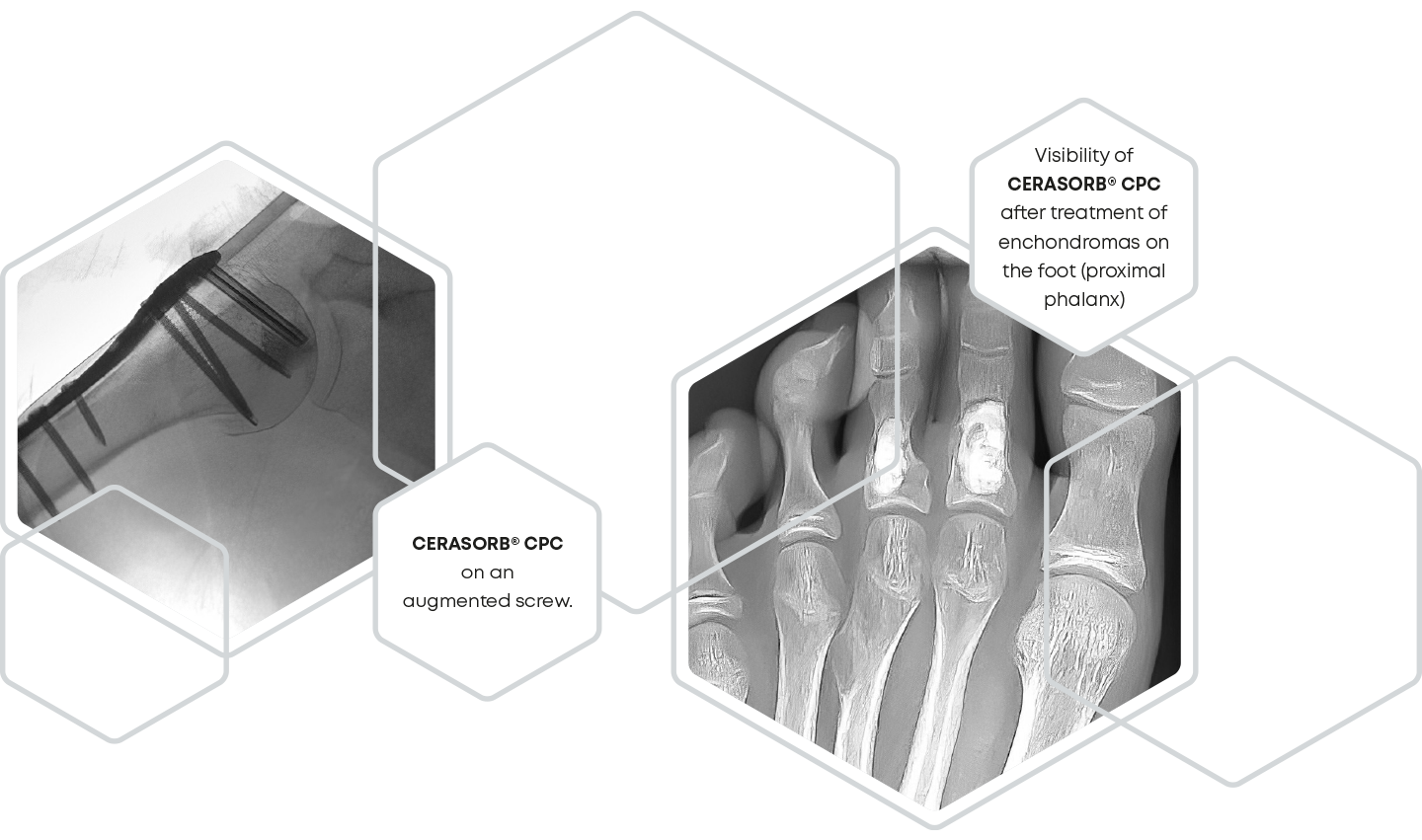
The release of particles during the setting reaction of a bone substitute cement is significantly influenced by the cohesion. Accordingly, during the development of CERASORB® CPC, care was taken to minimize particle release to below 2 % after injection in water. Thus, even in the case of screw augmentation, CERASORB® CPC fills the entire defect and remains completely in the bone cavity instead of flowing back along the screw from the tip to the head or leaking out under the screw head.
(For the complete list of indications and contraindications, please refer to the current version of the Instructions for use).
University Hospital Regensburg, Germany. Department of Trauma Surgery
Lateral tibial plateau fracture left tibia, type Schatzker V
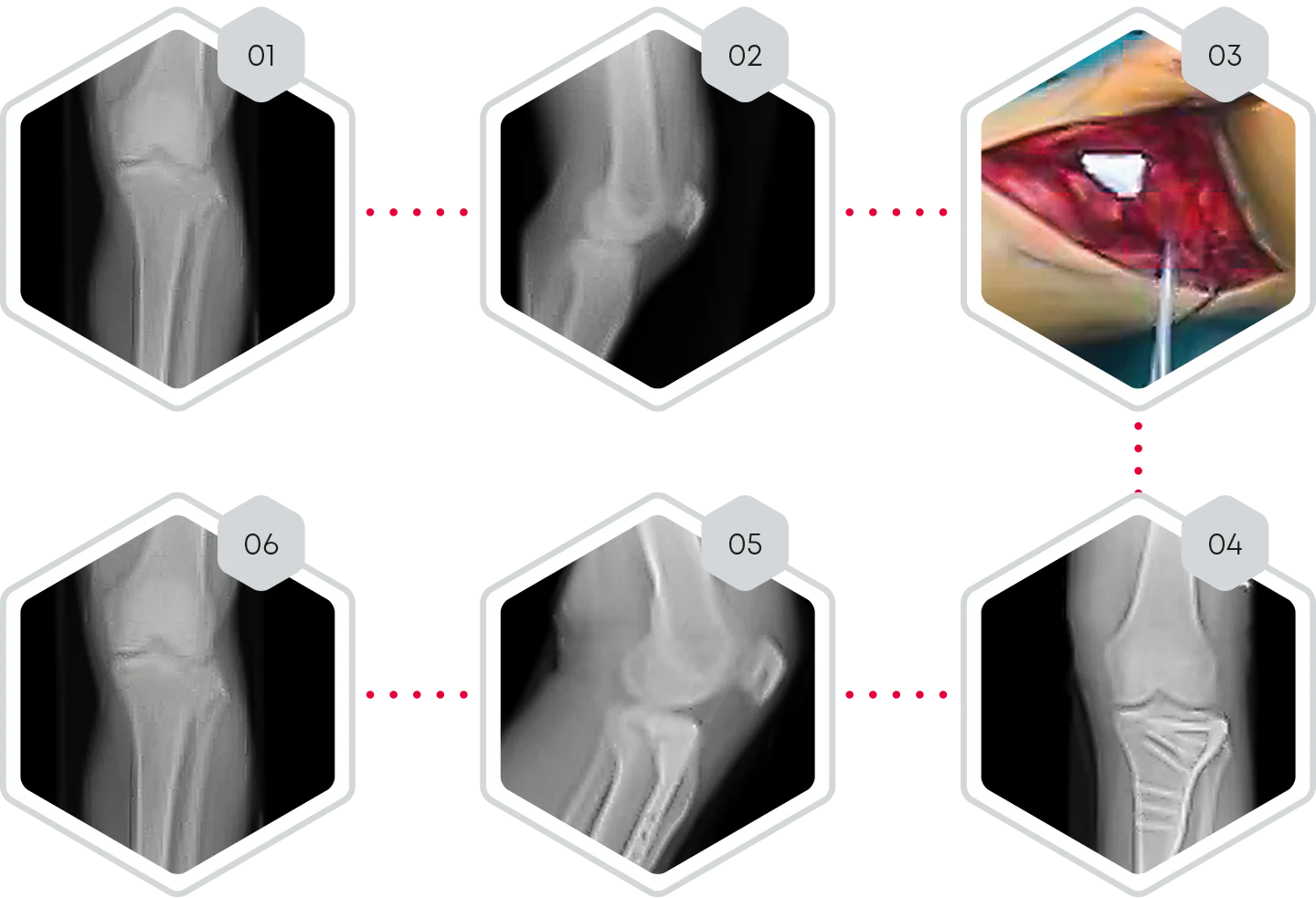
❯ 01-02 Pre-operative X-rays of tibial plateau fracture, type Schatzker V
❯ 03 Open reduction of lateral tibial plateau left and bone defect filling with CERASORB® CPC
❯ 04-05 Day 1 post-operative x-rays.
❯ 06 6 months post-operative x-rays.
❯ Clinical follow-up.
❯ CERASORB® CPC was easy to handle during the operation and allowed sufficient bone defect filling.
❯ 6 months post-operative, X-rays showed complete fracture healing with osseous integration and initial degradation of CERASORB® CPC. No subsidence of the articular surface could be observed.
❯ CERASORB® CPC supports the defect through good osseous integration and is gradually replaced by the newly formed bone.
University Hospital Regensburg, Germany. Department of Trauma Surgery
Ankle fracture type WEBER C with bony defect of distal fibula
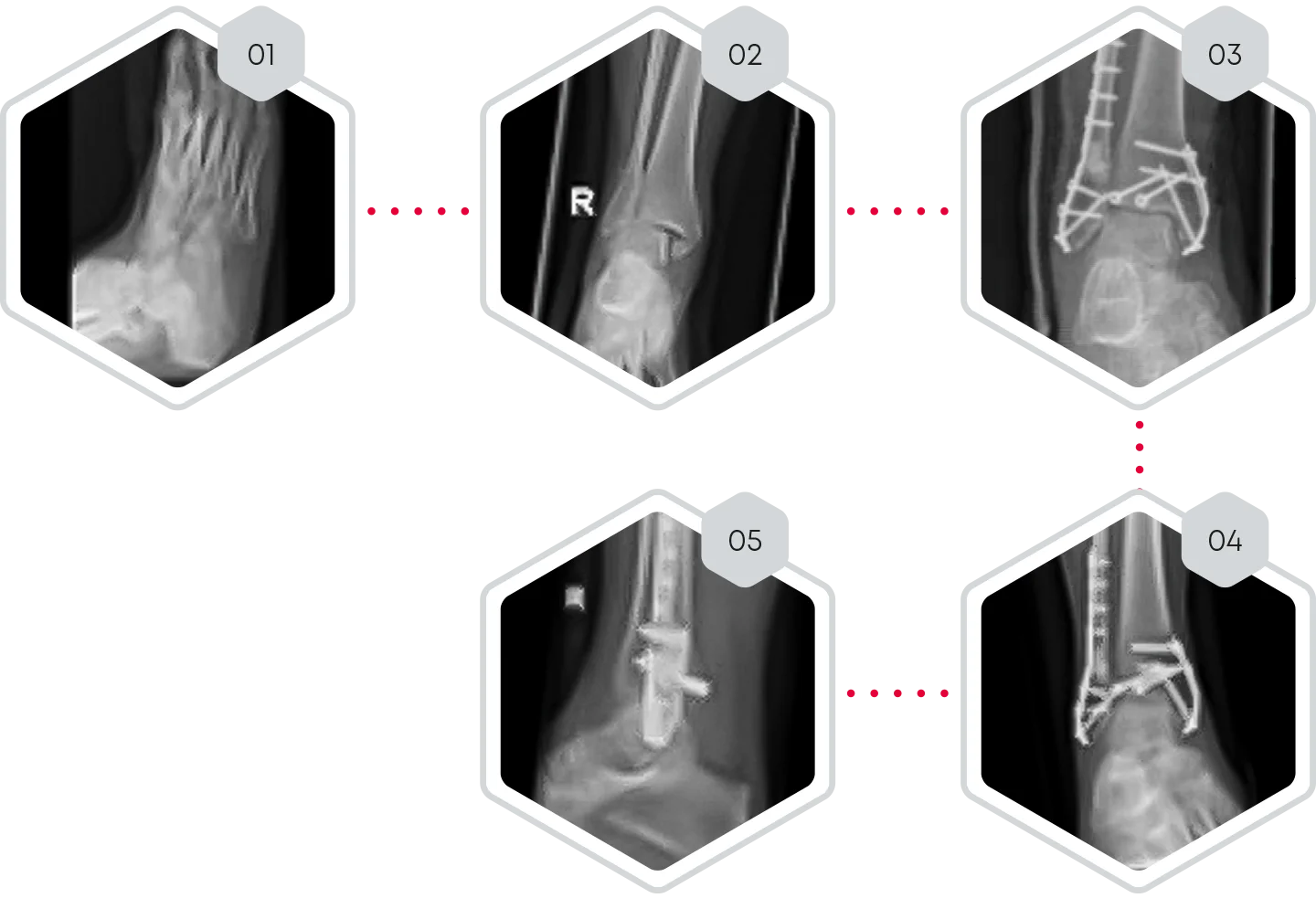
❯ 01-02 Pre-operative X-rays of ankle fracture type WEBER C with bony defect of distal fibula
❯ 03 Day 1 post-operative x-ray. Open reduction and filling of the fibula defect with CERASORB® CPC
❯ 04-05 10 weeks post- operative X-rays.
❯ Clinical follow-up.
❯ CERASORB® CPC was easy to handle during the operation
and allowed sufficient bone defect filling.
❯ 10 weeks post-operative, X-rays showed complete fracture healing
with beginning of osseous integration of CERASORB® CPC.
❯ CERASORB® CPC supports the defect through good osseous
integration and is gradually replaced by the newly formed bone.

❯ CERASORB® is a purely synthetic Bone Regeneration Material (BRM) with a phase purity of > 99% (contains less than 1% HA), which is referenced as the global ICDD gold-standard. There are no infection risks with CERASORB®.
❯ CERASORB® contains a well desinged and defined microporous sintering structure, that avoids particle break-up when used correctly. The primary particle size is < 63µm (d50= 10µm) to avoid phagocytosis and to be aligned with the natural bone resorption and remodelling ratio.
❯ The micro-, meso- and macropores of CERASORB® provide fast integration of vital cells, a high release of Calcium ions during resorption through hydrolytic corrosion and contribute to the transformation of osteoblasts to osteocytes.